
29.32 per 100 person-years hazard ratio 0.76, 95% confidence interval 0.68-0.84 GLY/IND vs. Risk of severe AE was lower among patients treated with UMEC/VI or GLY/IND than among those who received TIO/OLO (UMEC/VI vs. Baseline characteristics were well balanced after 1:1 matching of UMEC/VI and GLY/IND, 2:1 matching of UMEC/VI and TIO/OLO, and 2:1 matching of GLY/IND and TIO/OLO. Results Among the 44,498 patients identified and included, 15,586 received GLY/IND, 20,460 received UMEC/VI, and 8,452 received TIO/OLO. Propensity score matching and Cox regression models were used to compare outcomes of acute exacerbation (AE) and cardiovascular events associated with LAMA/LABA FDC treatment. Research Question What are the differences between available LAMA/LABA FDCs on the risk of acute exacerbation and cardiovascular events? Study Design and Methods This retrospective cohort study based on a national insurance claims database included COPD patients aged 40 and older who were newly prescribed glycopyrronium/indacaterol (GLY/IND), umeclidinium/vilanterol (UMEC/VI), or tiotropium/olodaterol (TIO/OLO) FDC between January 1, 2015, and June 30, 2019. 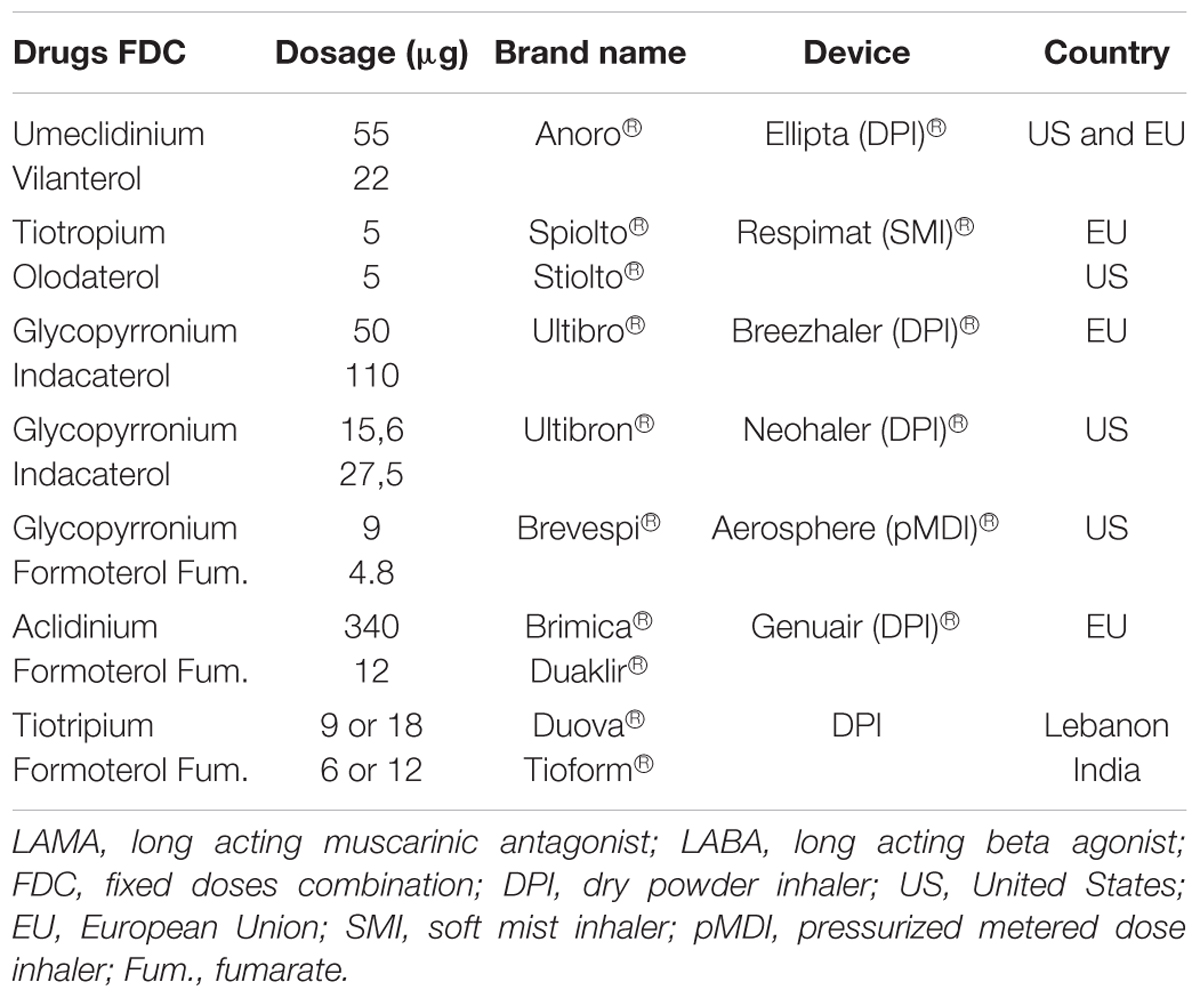
Further research will be required to confirm these findings.Ībstract = "ABSTRACT Background Researchers have yet to obtain conclusive evidence differentiating among fixed-dose combinations (FDCs) of long-acting muscarinic antagonists (LAMAs) and long-acting beta-agonists (LABAs) for chronic obstructive pulmonary disease (COPD) in terms of real-world clinical outcomes. Interpretation Our results revealed that the risk of severe AE was lower among COPD patients receiving UMEC/VI or GLY/IND than among those receiving TIO/OLO, while the incidence of cardiovascular events was similar across groups but was slightly lower in GLY/IND users when compared with TIO/OLO users. In addition, GLY/IND users tended to have a lower risk of cardiovascular events than TIO/OLO users, but the difference dissipated when restricting follow-up to a shorter duration. ABSTRACT Background Researchers have yet to obtain conclusive evidence differentiating among fixed-dose combinations (FDCs) of long-acting muscarinic antagonists (LAMAs) and long-acting beta-agonists (LABAs) for chronic obstructive pulmonary disease (COPD) in terms of real-world clinical outcomes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
